Toyota has long clung to the Nickel-Metal-Hydride (NiMH) batteries, claiming that lithium-ion batteries are too expensive. While that might be the case, there are dozens of research projects going on that aims to increase energy density, increase power density, and reduce the cost of batteries. What it means is that Toyota has primarily offered hybrid cars, where the other car companies have moved on to plug-in hybrids or all electric cars.
In the meantime Toyota is looking to solid state and lithium-air batteries. This is based to research information we find on the Toyota-Global website.
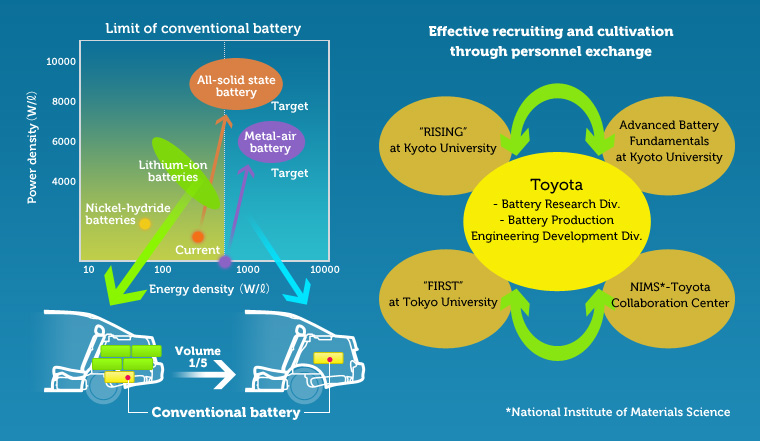
This first picture outlines a partnership between Toyota, Kyoto University and Tokyo University working on battery research, and advanced battery fundamentals.
The chart shows the current range of energy and power density, with NiMH providing energy density of slightly under 100 Watts/Liter and Power Density around 2000 Watts/Liter. Lithium-Ion is described in this chart as providing double that energy and power density. However both Solid State and “Metal-Air” (a.k.a. lithium-air) batteries providing far more.
The goal shown is a battery pack 1/5th the size of current battery packs. The effect of that goal is to preserve as much of the volume of the car for use of the humans, such as to carry more people or cargo.
The research began in 2010, so at this point (2013) it’s been going on for awhile.
The difference between current batteries and solid state batteries is that current batteries use a liquid electrolyte. If you think of the stereotypical 12 volt lead-acid battery in gas powered cars, it has caps covering cells that you must periodically fill with distilled water. That water serves as the electrolyte and acts as the medium for sending electrons back and forth between anode and cathode. In lithium-ion cells the liquid is much less liquid.
What Toyota has to say is: By modifying liquefied electrolytes into solid electrolytes, it allows
each cells to connect without the need for individual casing, which
results in creating a more compact packaging.
The picture tries to show this. The two sides have the same number of cells, for the same pack voltage. But the box on the right is smaller than the box on the left, meaning the box on the right has a higher energy/power density. This efficiency increase is coming from improved packaging.
What makes that possible is the use of solid electrolytes. Rather than a liquid sloshing around, a solid state battery has blocks of solid material pressed together. This requires less packaging to achieve the same effect as the liquid electrolyte batteries.
The last picture shows one reason why metal-air (lithium-air) batteries have further energy density gains from packaging. Regular batteries carry both anode and cathode inside themselves. With metal-air batteries, the cathode is “air”, meaning that the battery weight is only the anode.
The problem with this picture is that with current lithium-air battery research, the “air” must be pure oxygen, and cannot be any old air from the atmosphere. This is because of contaminants and especially humidity. Lithium-metal burns in certain conditions. To resolve this problem lithium-air battery researchers have gizmos to extract pure oxygen from the atmosphere to provide the proper stuff to the lithium-air battery. That gizmo adds to the packaging complexity of lithium-air batteries in a way that is not shown on Toyota’s picture.
Source: Research Progress : Next Generation Secondary Batteries ![]()
- Highway design could decrease death and injury risk, if “we” chose smarter designs - March 28, 2015
- GM really did trademark “range anxiety”, only later to abandon that mark - March 25, 2015
- US Government releases new regulations on hydraulic fracturing, that some call “toothless” - March 20, 2015
- Tesla Motors magic pill to solve range anxiety doesn’t quite instill range confidence - March 19, 2015
- Update on Galena IL oil train – 21 cars involved, which were the supposedly safer CP1232 design - March 7, 2015
- Another oil bomb train – why are they shipping crude oil by train? – Symptoms of fossil fuel addiction - March 6, 2015
- Chevron relinquishes fracking in Romania, as part of broader pull-out from Eastern European fracking operations - February 22, 2015
- Answer anti- electric car articles with truth and pride – truth outshines all distortions - February 19, 2015
- Apple taking big risk on developing a car? Please, Apple, don’t go there! - February 16, 2015
- Toyota, Nissan, Honda working on Japanese fuel cell infrastructure for Japanese government - February 12, 2015